“An ounce of prevention is worth a pound of cure.”
Ben Franklin once said that famous line and it has a lot of relevance to us physically active folks, especially to athletes and weightlifters.
People will rush to buy muscle builders, protein powders, hormone boosters, fat burners, and the like, but they often overlook strategies and products to improve general health and longevity, like preserving joint health.
Well, anybody who has spent some time in the gym training hard knows how vitally important healthy joints are.
More than 52 million Americans suffer from arthritis, and if your joints are inflamed or damaged, all aspects of your performance decline.
When your knees, hip, and shoulders ache, you lose speed, strength, power, and agility; you have to stop doing certain exercises and activities; you can’t handle weights you once could; and you often have to learn to deal with constant, nagging aches and pains.
Healthy joints are one of those things people just don’t fully appreciate until they’re gone.
Well, in this article, you’re going to learn the most common causes of joint pain and how to naturally and safely treat and prevent it.
What Causes Joint Pain?
Joints are places in the body where two bones connect and, in most cases, they provide structural support and allow for movement.
For example, your hip joint connects your femur to your pelvis, your knee joint connects your femur and your tibia, and your shoulder joint connects your shoulder blade to your arm (humerus).
Structurally speaking, there are three types of joints: fibrous, cartilaginous, and synovial joints.
- Fibrous joints are connected by a dense connective tissue called collagen. These joints are all called “fixed joints” because they don’t move.
Examples of fibrous joints are those found between the bones of the skull and between certain long bones in the body like the radius and ulna.
- Cartilaginous joints are connected by cartilage, a stiff, collagen-rich connective tissue. Cartilaginous joints allow for more movement than fibrous joints.
Examples of cartilaginous joints are the discs between your vertebrae and the first sternecostal joint, which is where your first rib connects to the breastbone.
- Synovial joints are the most common movable joints in mammals. They are connected by intricate structures consisting of a cavity filled with fluid, a capsule enveloping it, cartilage, and, depending on the joint, various other tissues like discs, pads, tendons, ligaments, and fluid-filled saclike structures (bursae) that further reduce friction.
That’s quite a mouthful to process, so here’s an image to help:
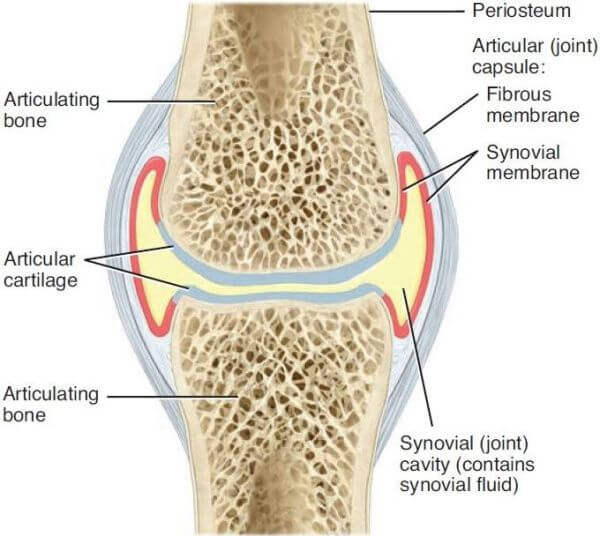
Examples of synovial joints are elbow joints, shoulder joints, wrist joints, knee joints.
What, then, causes the discomfort, aching, and soreness of joint pain?
Well, common causes include…
- Arthritis (which we’ll talk more about soon)
- Bursitis (inflammation of the bursae)
- Tendonitis (tendon inflammation)
- Tendon and/or ligament injury (strains and sprains)
- Muscle strains
- Bone injury (fractures and breaks)
- Dislocation
Let’s get more specific though and talk about the most common joint pains people struggle with.
What Causes Knee Pain?
Knee pain is widespread and especially prevalent among athletes.
Common causes for knee pain are, as noted above, strains and sprains, cartilage tears, and tendon inflammation, but the complex nature of the knee joint makes it susceptible to other problems.
For example…
- Repeated bending and kneeling can aggravate the fluid-filled sac over the kneecap.
- Illiotibial band syndrome, which is pain caused by the aggravation of the illiotibial (IT) band–a swath of fibrous tissue (fascia) that extends from your hip to the outer part of your knee.
- Muscle imbalances and tightness and misalignment of the legs.
- Inflammation of the tendons surrounding your knee (patellar tendonitis is particularly common).
What Causes Hip Pain?
Like with knee pain, the most common causes of hip pain are arthritis, bursitis, tendonitis. Muscle imbalances are quite common as well.
One of areas of the hip joint that often gives athletes problems is the sacroiliac joint in the pelvis.
Here’s what it looks like:
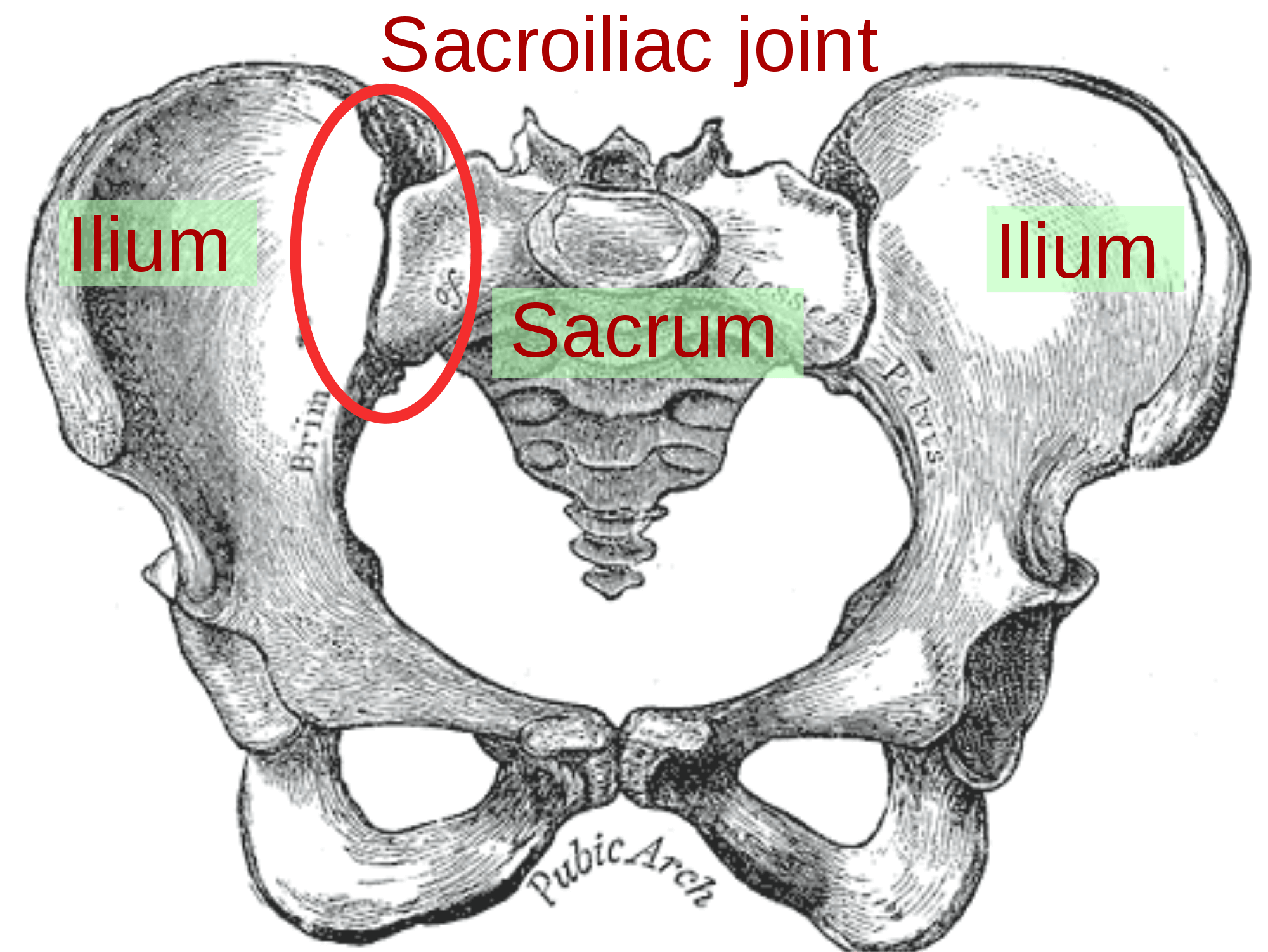
The sacroiliac joint is designed to transfer forces to and from the lower spine into the hip and legs and when it gets aggravated or injured, pain is felt in the lower back, hip, and/or legs.
The squat motion puts a tremendous amount of pressure on the sacroiliac joint, which is one of the reasons why many weightlifters in particular struggle with sacroiliac pain.
What Causes Shoulder Pain?
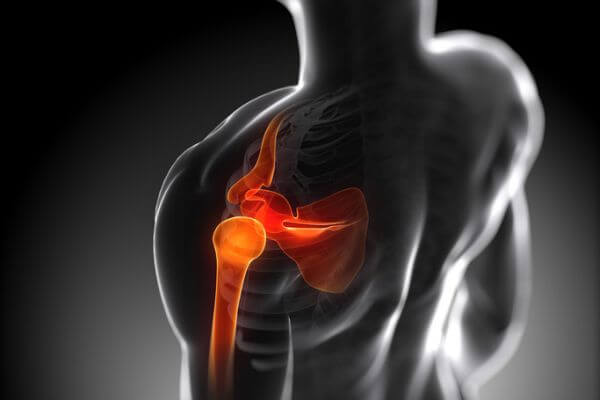
The shoulder is an intricate, extremely mobile (and rather amazing, really) joint.
It moves the shoulder forward and backward and allows the arm to move in a circular motion as well as up and away from the body.
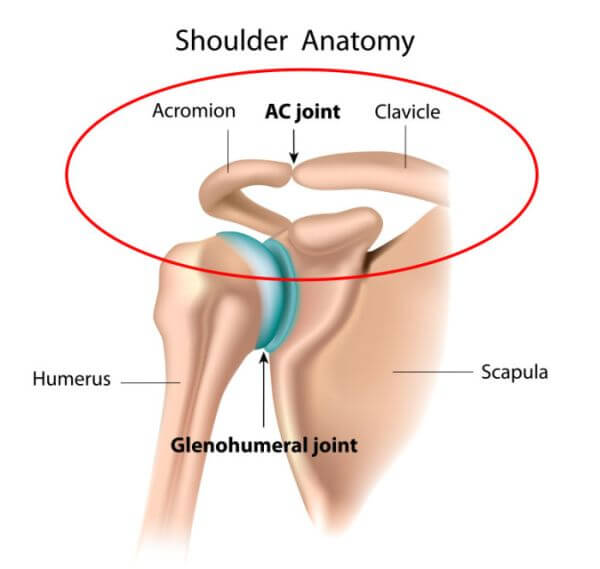
The structure of the shoulder joint is a “ball-and-socket” design with three main bones cushioned by a layer of cartilage: the humerus (the long bone running from your shoulder to your elbow), clavicle (collarbone), and scapula (shoulder blade).
There are two main joints in the shoulder:
- The the acromioclavicular joint. This connects the highest part of the scapula– the acromion–and the clavicle.
- The glenohumeral joint. This is comprised of the ball-shaped top of the humerus and the socket on the scapula.
Here’s how they look:
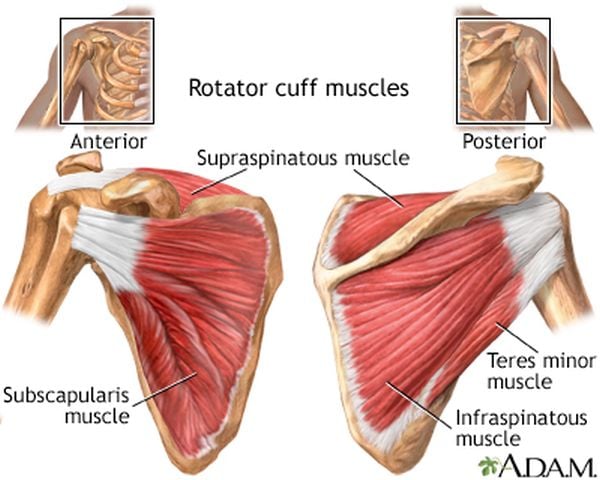
The shoulder’s impressive range of motion is produced and regulated by the rotator cuff, which is comprised of four muscles and their tendons: the supraspinatus, infraspinatus, teres minor, and subscapularis.
Here’s a visual:
Rotator cuff injuries are more common among weightlifters than the rest of the population for a couple of reasons:
- Many people, and guys in particular, do a lot of bench pressing and shoulder pressing and not enough pulling. This causes an imbalance between the anterior and posterior muscle groups in the torso.
- Many people also bench press with improper form, which puts the rotator cuff muscles in a compromised, injury-prone position.
What is Arthritis?
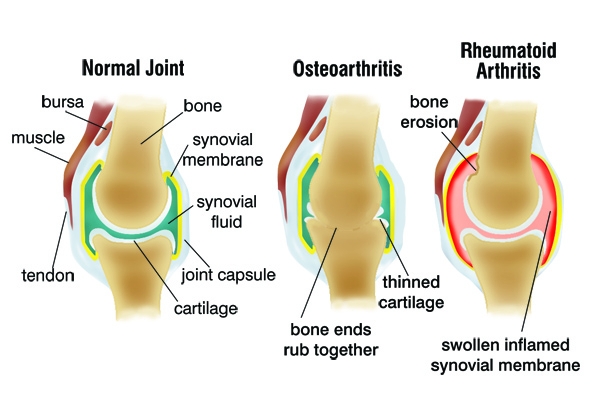
Many people mistakenly think that arthritis is something unavoidable and inevitable–that we will all succumb to at some point in our lives.
Well, while we do have to accept our bodies will eventually wear out, if we take good care of our joints, they can remain healthy and functional for a long, long time.
As you know, your joints are lined with the flexible tissue cartilage, which lubricates them and absorbs physical impacts. Healthy cartilage preserves ease of motion and keeps your joints pain free.
Many people assume that regular exercise must accelerate cartilage loss through repetitive use and general wear and tear. Research shows otherwise, though.
Forms of exercise usually assumed to be harmful to your joints like weightlifting and long-distance running are not associated with cartilage loss. In fact, regular exercise appears to help preserve healthy cartilage levels.
Why, then, do so many long-term exercisers and athletes seem to suffer from joint problems? And what can be done about it?
Well, to answer those questions, let’s take a closer look at how joints lose their structural integrity, eventually resulting in arthritis.
Two common forms of arthritis are osteoarthritis and rheumatoid arthritis, which involve painful inflammation of the joints resulting from the gradual loss of the cartilage.
Osteoarthritis has been long believed to be a natural consequence of aging, and rheumatoid arthritis is an autoimmune disease that results in joint inflammation and destruction.
Well, we now know that both conditions are at least partially caused by an unwanted destructive immune response to joint collagen that eats away at the joints. What happens is your body’s immune system incorrectly targets and attacks joint cartilage as a foreign, unwanted substance.
This can become a vicious cycle: the assault slowly destroys the joints, which causes even more inflammation, which triggers a larger immune response.
Theoretically, then, if we could stop this internal assault on your joints as well as reduce existing joint inflammation, we could dramatically improve your joint health and reduce the risk of disease and dysfunction, including arthritis.
And that’s exactly what research suggests.
Studies show that, in people with arthritic conditions, reducing inflammation and “teaching” the immune system to stop attacking proteins found in joint cartilage can significantly improve joint health and function and decrease or even eliminate pain and swelling.
There are several supplements proven to help with this. We’ll talk more about them soon.
Is Weightlifting Bad For Your Joints?

The assumption that weightlifting will degrade your joints over time makes sense at first glance.
When you’re squatting, pushing, and pulling hundreds of pounds week after week, you have to wonder how healthy it is for your joints.
Fortunately, research doesn’t support these fears.
For example, this study was conducted a few decades ago with 25 competitive weightlifters.
These subjects spent a lot more time training and lifted a lot more weight than you or I probably do, and researchers were surprised to find that on the whole, their joints were as healthy or even healthier than other people their age.
Furthermore about half of the subjects admitted they were using anabolic steroids, which means their joints were subjected to loads beyond what would be normally possible.
It’s worth noting that the Olympic lifters had the most joint problems out of the group. This isn’t surprising given the risky nature of these movements and the sheer amount of weight that competitive lifters move around.
One other point worth noting is researchers found that previously injured joints were more susceptible to joint degeneration than healthy joints. So if you’ve sustained joint injuries in the past, lifting heavy weights week after week may cause them to get worse.
These findings are in line with other research conducted specifically on the squat and deadlift, which are two of the most maligned and, ironically, effective, exercises you can do.
- Even in extreme cases of squatting, such as powerlifters lifting 2.5 times bodyweight, the compressive forces placed on the knee and its tendons are well within its ranges of ultimate strength.
In one study, the highest ACL force recorded when squatting was a mere 6% of its ultimate strength). Highest recorded PCL forces were well within natural strength limits as well.
There’s quite a bit of research that suggests that strength training, and squatting in particular, is an effective treatment for osteoarthritis, both in terms of reducing pain and improving function.
- Research conducted by scientists at the University of Waterloo used real-time x-ray imaging to watch the spines of elite powerlifters while they fully flexed their spines with no weights and while they deadlifted over 400 pounds.
With the exception of one subject, all men completed their deadlifts within the normal range of motion during full flexion. Ligament lengths were unaffected, indicating that they don’t help support the load but instead limit range of motion.
A study conducted by researchers at the University of Valencia found that the deadlift is an extremely effective way to train the paraspinal muscles, which run down both sides of your spine and play a major role in the prevention of back injuries.
So then, if weightlifting isn’t inherently bad for your joints, why do so many weightlifters seem to have shoulder, knees, and lower back problems?
How to Ruin Your Joints With Weightlifting

Despite common misconceptions, weightlifting just isn’t a very dangerous activity. You’re far more likely to get injured playing just about any sport than you are lifting weights.
That said, like with any physical activity, there’s always a risk of injury and the occasional ache or strain is inevitable.
Well, when it comes to weightlifting, the two best ways to get hurt is…
- try to lift too much weight; and…
- train with poor form.
These are the cardinal sins of weightlifting, and are what give it and the big, compound exercises a bad reputation.
You see, weightlifting just isn’t a forgiving activity if you don’t “respect” it. Get sloppy with heavy weights and bad things can happen. This doesn’t just apply to free weights either–machines can be just as “dangerous” when used improperly.
So, take the time to learn proper form and leave your ego at the door when you lift, and you’ll dramatically decrease your chance of getting hurt.
Go for that extra rep on your squat or deadlift…but don’t turn into a hunchback or turn the exercise into a Good Morning. Try to hit that PR on your bench press…but don’t flare your elbows to your ears.
Another way to get hurt is to ignore pain.
“No pain, no gain,” right? Not really.
Muscle soreness and the “burn” caused by lactic acid are very different than pain. Pain means something is wrong and trying to muscle through it is asking for trouble.
The key to dealing with pain is first, stopping whatever you’re doing, and second, treating it like an injury until it’s better.
Avoid exercises that aggravate the affected area, apply ice and heat accordingly, and let it heal.
If that means no deadlifting or squatting for a few weeks, so be it. Find alternative exercises that you can do pain free.
And trust me, I know that backing off while your body heals can be annoying. But hurting yourself and losing months of progress is much more frustrating.
5 Effective Joint Pain Remedies You Need to Try
Ask the wrong “expert” about what you should do to relieve your joint pain and you’re going to hear drugs or surgery or both.
The problems with anti-inflammatory drugs are they just mask the problem and long-term use is a bad idea.
The problem with surgery is obvious: it’s a traumatic, risky experience that we would all rather avoid if at all possible.
Well, short-term use of drugs can give some relief and some situations necessitate surgery but if yours doesn’t (and a good sports doc can tell you), there’s a good chance the following 5 pain-relief strategies can help.
Mobility Exercises
“Unsticking” soft tissues (muscles, tendons, ligaments, skin, and fascia) and improving movements patterns and range of motion can be very effective in relieving joint pain.
The type of exercises that accomplish this are commonly referred to as “mobility exercises,” and the right ones can work wonders.
Check out this article to learn more about how to use mobility exercises to reduce or even eliminate your joint pain.
Rest, Ice, and Heat
The most important part of recovery is rest.
That doesn’t mean immobility but it does mean you have to not do things that are going to impair healing and recovery.
Violate this simple principle and injuries can become chronic and debilitating.
Once a joint has fully healed and you’re ready to start training again, it’s a good idea to start with lighter weights and see how you feel over the next several days (no pain is a good sign), and gradually work back into your normal routine.
Ice helps you recover by reducing inflammation and swelling as well as internal bleeding from injured capillaries and blood vessels.
As long as there is pain and inflammation, ice will help.
Don’t apply ice for more than 15 to 20 minutes at a time, but you can rotate on and off all day.
Heat stimulates blood flow, which helps your body bring nutrients to and remove waste products from the area faster.
Heat shouldn’t be introduced immediately following an injury, however.
The general advice is to ice only for the first 3 days to bring down swelling and, once this has been accomplished, introduce heat. Alternate between 15 to 20 minutes of ice and heat.
Acupuncture
As you know, abnormalities in soft tissues can cause joint pains, and research shows acupuncture can help treat this.
Specifically, needling can help release “trigger points” in the body, which are tight, painful areas of muscle that refer pain to other areas of the body.
For example, when you press on a trigger point in your neck, you might also feel pain in your shoulder (I’ve personally experienced that one).
The key here is obviously the skill and knowledge of the acupuncturist. Look for someone trained in using acupuncture for myofascial release.
Massage
Like acupuncture, research shows that massage is another effective strategy for releasing trigger points.
This can not only relieve pain but can help prevent muscular problems from developing in the first place that can, in time, cause joint pains.
Arnica Gel
Arnica gel is produced from the Arnica Montana plant and has long been used to treat inflammation and pain in the muscles and joints.
Research shows that arnica gel can be an effective way to reduce joint pain. In fact, it can be equally as effective as ibuprofen in alleviating joint pains associated with arthritis.
Here’s the product I use when I have joint pain:
Joint Supplements That Do and Don’t Work
Bone and joint health supplements comprise a multi-billion dollar market that is only getting bigger and bigger.
When people are in pain and looking for a solution, they’ll buy and try just about anything.
Before you jump on the joint supplement bandwagon, though, let’s go over your options.
Glucosamine
If you’re boning up on joint health (har har har), you’ve heard about glucosamine.
It’s a supplement derived from shellfish and is often sold as a sure bet for relieving joint pain and improving joint health.
Well, studies show that it can provide minor pain relief but is unreliable in this mechanism.
Research also shows that glucosamine can reduce the rate of collagen loss, which is particularly relevant to people that run or participate in other high-impact activities regularly. Its collagen-preserving effects are relatively weak, though.
The bottom line is glucosamine isn’t worthless but also isn’t nearly as effective as many supplement companies claim.
It’s not the most popular joint supplement because it’s the best. It’s just cheap to produce (great profit margins) and heavily marketed.
If you want to get the absolute most out of a joint supplementation regimen and don’t have a budget, it makes sense to include glucosamine in it.
The clinically effective dosage of glucosamine is 900 to 1,500 mg per day, and here’s the product I personally use:
Chondroitin
Chondroitin is an major constituent of collagen and is frequently paired with glucosamine to treat the symptoms of arthritis.
Research casts doubt on its effectiveness, though.
Based on the scope and quality of the more recent research on chondroitin, my recommendation is save your money.
Methylsulfonylmethane (MSM)
MSM is a sulfur-containing chemical found in plants, animals, and humans. It’s often paired with glucosamine because of its antioxidative and anti-inflammatory properties.
Studies show that MSM has promise for treatment of arthritis but more rigorous research is needed.
Many people report that MSM relieves their joint pain so, like glucosamine, I think it’s worth including if budget isn’t a concern.
The clinically effective dosage of MSM appears to be around 3,000 mg per day, and again, here’s the product I personally use:
Curcumin
Curcumin is the yellow pigment found in the turmeric plant, which is the main spice in curry. It has been used therapeutically in Ayurvedic medicine for thousands of years.
Its health benefits are extensive, and scientific researchers around the world are investigating applications for fighting a variety of diseases such as cancer, cardiovascular disease, osteoporosis, diabetes, Alzheimer’s, and more.
One reason for this is that curcumin has powerful anti-inflammatory effects, which are exerted by inhibiting proteins that trigger the production of inflammatory chemicals.
Curcumin has a significant downside, however: intestinal absorption is very poor. So much so that supplementation without enhancement more or less eliminates the majority of its health benefits.
Fortunately, there’s an easy solution for increasing bioavailability: black pepper extract.
Research shows that pairing black pepper extract with curcumin increases bioavailability twentyfold. And when you do that, curcumin becomes an effective joint support supplement.
Studies show that supplementation with curcumin and black pepper extract reduces inflammatory signals in the joints and, in those with arthritis, relieves pain and stiffness and improves mobility.
When paired with piperine, the clinically effective dosages of curcumin range between 200 and 500 milligrams.
Given its powerful anti-inflammatory effects and long list of additional health benefits, curcumin is on my “highly recommended” list for both joint and general health.
And that’s why I included 500 mg of curcumin and 25 mg of piperine in each serving of my joint supplement, FORTIFY.
That’s only the beginning of what FORTIFY has to offer, though.
It also contains clinically effective dosages of three more molecules shown to alleviate joint pain and improve joint health.
Let’s take a look at each.
Undenatured Type II Colagen
Collagen is the main protein of the various connective tissues in animals and type II collagen (CII) is a type of collagen that makes up your joint cartilage.
“Undenatured” is more than a fancy word—it’s vitally important to the effectiveness of the product.
Denaturization is the alteration of the natural structure of a substance, usually by the addition of another chemical or heat that changes the substance’s physical properties.
Typical commercial processing of collagen causes alterations to its basic form (denaturization), and research shows that denatured collagen has no beneficial effects on joint inflammation.
Undenatured collagen, however, is a more natural form of the substance and research shows that the UC II® undenatured type II collagen is highly effective for regulating the immune response that inflames joints and destroys cartilage and bone, which further inflames the joints and starts a degenerative cycle.
And the best part about undenatured type II collagen is that these effects have been demonstrated in people with arthritic conditions and people with healthy joints.
It accomplishes this by “teaching” the body’s immune system to stop attacking collagen as a foreign substance, which is the cause of some arthritic conditions. Orally ingesting CII works almost like a vaccine, resulting in less of an inflammatory response to your own collagen because the body now recognizes it.
The clinically effective dosages of undenatured type II collagen range between 10 and 40 milligrams.
Undenatured type II collagen isn’t cheap but research shows it’s one of the most effective supplements you can take for preserving your joint health. I highly recommend it.
That’s why my joint supplement FORTIFY contains 10 milligrams of undenatured type II collagen per serving.
I chose the lower end of the clinically effective dosage because it isn’t clear if more is better. Research clearly shows that 10 mg is effective but not that two, three, or four times that amount is more so.
Boswellia Serrata
Boswellia serrata is a plant that produces an aromatic substance known as frankinsence, which has been used for thousands of years in Ayurvedic medicine to treat various disorders related to inflammation.
Thanks to modern science, we now know why.
Frankinsence contains molecules known asboswellic acids. Research shows that, like curcumin, boswellic acids—and one in particular known as acetyl-keto-beta-boswellic acid, or AKBA—inhibit the production of several proteins that cause inflammation in the body.
And in case you’re wondering, the difference between the anti-inflammatory mechanisms of curcumin and boswellic acids is they work on different enzymes. Curcumin inhibits an enzyme known as cyclooxygenase, or COX, and boswellic acids inbhibit lysyl oxidase, or LOX (and, most notably, 5-LOX).
These anti-inflammatory properties extend to the joints, which is why studies show that Boswellia serrata is an effective treatment for reducing joint inflammation and pain as well as inhibiting the autoimmune response that eats away at joint cartilage and eventually causes arthritis.
The clinically effective dosages of boswellia serrata range between 100 and 200 milligrams.
What can I say…I’m a fan of this molecule. Its powerful anti-inflammatory effects are not only good for your joints but for your entire body, as systemic inflammation is a known contributor to many types of disease.
FORTIFY contains 125 mg of boswellia serrata per serving.
Grape Seed Extract
Grape seed extract is a substance derived from the ground-up seeds of red wine grapes, which have been used in European medicine for thousands of years.
There are two molecules in grape seed extract that account for most of its health benefits: tannins, which are bitter compounds that make wine taste dry, and procyanidins, which are chains of antioxidants found in some plants.
Much of the research on grape seed extract’s beneficial effects on joint health is extrapolated from research on a similar molecule known as pycnogenol, which is also a potent source of procyanidins. Pycnogenol is significantly more expensive than grape seed extract, however, which is why GSE is preferable for supplementation needs.
Research shows that the primary way grape seed extract helps protect joint cartilage from damage caused by the pro-inflammatory immune response that can develop into arthritis.
Studies also show that supplementation with grape seed extract confers other health benefits, including the following:
- Better eye health.
- A reduced risk of heart disease.
- Greater blood glucose control.
- Improved blood flow to the extremities, which can reduce the appearance of varicose veins.
- Potential anti-cancer activities.
Clinically effective dosages of grape seed extract range from 75 to 300 mg.
You get a lot for your money with grape seed extract. Like curcumin and boswellia serrata, it improves both joint and overall health, and is a worthwhile inclusion in your supplementation regimen.
Like curcumin and boswellia serrata, grape seed extract improves both joint and overall health, and is a worthwhile inclusion in your supplementation regimen.
FORTIFY contains 90 mg of grape seed extract per serving.
Spirulina
Spirulina is a non-toxic blue-green algae that is very similar to fish oil in its health benefits.
Research has shown that supplementation with spirulina can…
- Reduce muscle damage caused by exercise
- Improve exercise performance
- Increase strength
- Improve cholesterol and triglyceride levels
- Reduce blood pressure
- Improve blood sugar control
- Reduce systemic inflammation
- Improve allergy symptoms
- Improve insulin sensitivity
The anti-inflammatory effects are what help with reducing joint pain, and animal research has shown that spirulina supplementation improves joint health.
Spirulina has a lot to offer, which is why it’s in my cabinet and on my list of supplements everyone should take. Here’s the product I use:
Fish Oil
The next type of general health supplement that I highly recommend is fish oil, because it’s a great source of “omega-3 fatty acids.”
Omega-3 fatty acids (eicosapentaenoic acid–EPA–and docosahexaenoic acid–DHA) are an essential type of fat, meaning they can’t be synthesized by the body and must be obtained from the diet.
Research shows that fish oil has powerful anti-inflammatory effects and can significantly reduce joint pain.
Studies also show that supplementation with fish oil can…
- Increase muscle protein synthesis
- Reduce muscle soreness, inflammation, and anxiety
- Reduce blood pressure, depression, the negative effects of stress, and the risk for kidney and cardiovascular disease, as well as stroke and metabolic syndrome
- Improve glucose uptake and insulin sensitivity in people with impaired insulin response and metabolism, and preserve it in the metabolically healthy
- Improve memory and cognitive performance
- Help prevent weight gain
- Speed up fat loss
If you don’t get enough omega-3 fatty acids in your diet (2 to 3 grams per day is a good target), I strongly suggest that you take a fish oil supplement.
Not all fish oils are made the same, though. There are two important things to consider when choosing one:
You want to know how the oil has been processed.
There are two forms of fish oil on the market today: the triglyceride form, and the ethyl ester form.
The triglyceride form is fish oil in its natural state, and the ethyl ester form is a processed version of the triglyceride form that includes a molecule of ethanol (alcohol).
While plenty of studies have proven the benefits of supplementation with fatty acid ethyl esters (FAEEs), research has shown that the triglyceride form is better absorbed by the body.
One of the reasons for this is the ethyl ester form is much more resistant to the enzymatic process by which the body breaks the oil down for use.
Another downside to the ethyl ester form is during the digestive process, your body converts it back to the triglyceride form, which results in the release of the ethanol molecule.
Although the dose is small, those with alcohol sensitivity or addiction can be negatively affected. Furthermore, research has provided evidence of cellular and organic toxicity and injury resulting from the ingestion of FAEEs
You want to know the EPA/DHA content of each serving.
Because of the varying quality of fish oils on the market, it’s important that you look at how many milligrams of EPa and DHA (omega-3 fatty acids) are actually in each serving.
Lower quality supplements might have as little as 150 – 200 milligrams per 1 gram of fat, which makes them nearly worthless as you have to take far too much every day to get enough omega-3s (you want a minimum of 2 – 3 grams of omega-3s per day).
A high-quality fish oil can be quite a bit more money than a low-quality one, but when you look at how much you’re getting for that money in terms of omega-3 fatty acids, the price makes more sense.
For example, here’s the label from a cheap, low-quality (ethyl ester) fish oil product:
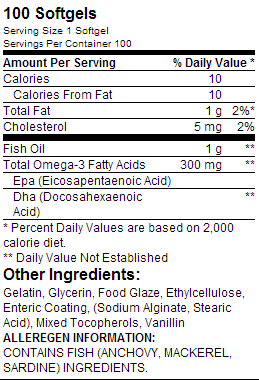
This product costs about $11, and comes with 100 pills containing 300 mg of omega-3 fatty acids each. This means you’re getting 30 grams of omega-3 fatty acids per bottle, and paying about 37 cents per gram.
Now, here’s the label from a high-quality triglyceride fish oil product that I use, Nordic Naturals’ Ultimate Omega:
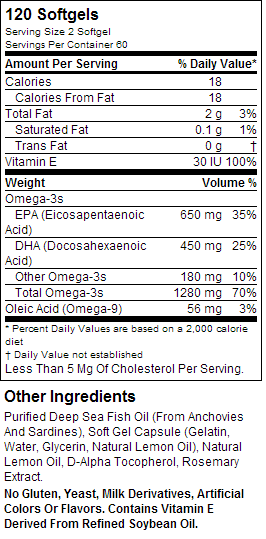
This product costs about $40, and comes with 120 pills containing 640 mg of omega-3 fatty acids each.
This means you’re getting about 77 grams of omega-3 fatty acids per bottle, and paying about 52 cents per gram.
So, as you can see, the initial price difference of $11 vs. $40 isn’t as drastic when you look at what you’re getting:
37 cents per gram of low-quality oil that isn’t likely to deliver all of the benefits you’re looking for vs. 52 cents per gram for the highest quality oil on the market that will.
Thus, I recommend you go with the high-quality product whose nutrition facts label I showed above:
The Bottom Line on Joint Pain
Joint pain is one of those things that people tend to ignore or mask with drugs, usually without realizing that negligence can lead to chronic disease or dysfunction.
It’s unfortunate too because, in many cases, it doesn’t take too much to resolve joint pain and prevent it from reoccurring.
My personal routine is simple:
- A series of mobility exercises for my shoulders and lower body.
- A weekly (rather uncomfortable) massage working out tight and painful “muscle knots.”
- Rest, ice, arnica, and heat when necessary.
- Supplementation with glucosamine and MSM, FORTIFY, spirulina, and fish oil.
This routine keeps my joints pain free and functional and I hope it can do the same for you.
What’s your take on joint pain? Have anything else to share? Let me know in the comments below!
Scientific References +
- Smith, G. I., Atherton, P., Reeds, D. N., Mohammed, B. S., Rankin, D., Rennie, M. J., & Mittendorfer, B. (2011). Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clinical Science, 121(6), 267–278. https://doi.org/10.1042/CS20100597
- Tartibian, B., Maleki, B. H., & Abbasi, A. (2009). The Effects of Ingestion of Omega-3 Fatty Acids on Perceived Pain and External Symptoms of Delayed Onset Muscle Soreness in Untrained Men. Clinical Journal of Sport Medicine, 19(2), 115–119. https://doi.org/10.1097/JSM.0b013e31819b51b3
- Bloomer, R. J., Larson, D. E., Fisher-Wellman, K. H., Galpin, A. J., & Schilling, B. K. (2009). Effect of eicosapentaenoic and docosahexaenoic acid on resting and exercise-induced inflammatory and oxidative stress biomarkers: A randomized, placebo controlled, cross-over study. Lipids in Health and Disease, 8. https://doi.org/10.1186/1476-511X-8-36
- Kiecolt-Glaser, J. K., Belury, M. A., Andridge, R., Malarkey, W. B., & Glaser, R. (2011). Omega-3 supplementation lowers inflammation and anxiety in medical students: A randomized controlled trial. Brain, Behavior, and Immunity, 25(8), 1725–1734. https://doi.org/10.1016/j.bbi.2011.07.229
- Ramel, A., Martinez, J. A., Kiely, M., Bandarra, N. M., & Thorsdottir, I. (2010). Moderate consumption of fatty fish reduces diastolic blood pressure in overweight and obese European young adults during energy restriction. Nutrition, 26(2), 168–174. https://doi.org/10.1016/j.nut.2009.04.002
- Sublette, M. E., Ellis, S. P., Geant, A. L., & Mann, J. J. (2011). Meta-analysis of the effects of Eicosapentaenoic Acid (EPA) in clinical trials in depression. Journal of Clinical Psychiatry, 72(12), 1577–1584. https://doi.org/10.4088/JCP.10m06634
- Hamazaki, T., Itomura, M., Sawazaki, S., & Nagao, Y. (2000). Anti-stress effects of DHA. BioFactors, 13(1–4), 41–45. https://doi.org/10.1002/biof.5520130108
- Simopoulos, A. P. (2008). The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. In Experimental Biology and Medicine (Vol. 233, Issue 6, pp. 674–688). Exp Biol Med (Maywood). https://doi.org/10.3181/0711-MR-311
- He, K., Rimm, E. B., Merchant, A., Rosner, B. A., Stampfer, M. J., Willett, W. C., & Ascherio, A. (2002). Fish consumption and risk of stroke in men. Journal of the American Medical Association, 288(24), 3130–3136. https://doi.org/10.1001/jama.288.24.3130
- Huang, T., Bhulaidok, S., Cai, Z., Xu, T., Xu, F., Wahlqvist, M. L., & Li, D. (2010). Plasma phospholipids n-3 polyunsaturated fatty acid is associated with metabolic syndrome. Molecular Nutrition and Food Research, 54(11), 1628–1635. https://doi.org/10.1002/mnfr.201000025
- Huang, T., Wahlqvist, M. L., Xu, T., Xu, A., Zhang, A., & Li, D. (2010). Increased plasma n-3 polyunsaturated fatty acid is associated with improved insulin sensitivity in type 2 diabetes in China. Molecular Nutrition and Food Research, 54(SUPPL. 1). https://doi.org/10.1002/mnfr.200900189
- Haugaard, S. B., Vaag, A., Mu, H., & Madsbad, S. (2009). Skeletal muscle structural lipids improve during weight-maintenance after a very low calorie dietary intervention. Lipids in Health and Disease, 8. https://doi.org/10.1186/1476-511X-8-34
- Narendran, R., Frankle, W. G., Mason, N. S., Muldoon, M. F., & Moghaddam, B. (2012). Improved Working Memory but No Effect on Striatal Vesicular Monoamine Transporter Type 2 after Omega-3 Polyunsaturated Fatty Acid Supplementation. PLoS ONE, 7(10), e46832. https://doi.org/10.1371/journal.pone.0046832
- Muldoon, M. F., Ryan, C. M., Sheu, L., Yao, J. K., Conklin, S. M., & Manuck, S. B. (2010). Serum phospholipid docosahexaenonic acid is associated with cognitive functioning during middle adulthood. Journal of Nutrition, 140(4), 848–853. https://doi.org/10.3945/jn.109.119578
- Buckley, J. D., & Howe, P. R. C. (2009). Anti-obesity effects of long-chain omega-3 polyunsaturated fatty acids. In Obesity Reviews (Vol. 10, Issue 6, pp. 648–659). Blackwell Publishing Ltd. https://doi.org/10.1111/j.1467-789X.2009.00584.x
- Couet, C., Delarue, J., Ritz, P., Antoine, J. M., & Lamisse, F. (1997). Effect of dietary fish oil on body fat mass and basal fat oxidation in healthy adults. International Journal of Obesity, 21(8), 637–643. https://doi.org/10.1038/sj.ijo.0800451
- Offman, E., Marenco, T., Ferber, S., Johnson, J., Kling, D., Curcio, D., & Davidson, M. (2013). Steady-state bioavailability of prescription omega-3 on a low-fat diet is significantly improved with a free fatty acid formulation compared with an ethyl ester formulation: The ECLIPSE II study. Vascular Health and Risk Management, 9(1), 563–573. https://doi.org/10.2147/VHRM.S50464
- https://pubmed.ncbi.nlm.nih.gov/24124374/
- Best, C. A., & Laposata, M. (2003). Fatty acid ethyl esters: toxic non-oxidative metabolites of ethanol and markers of ethanol intake. In Frontiers in bioscience : a journal and virtual library (Vol. 8). Front Biosci. https://doi.org/10.2741/931
- Remirez, D., González, R., Merino, N., Rodriguez, S., & Ancheta, O. (2002). Inhibitory effects of Spirulina in zymosan-induced arthritis in mice. Mediators of Inflammation, 11(2), 75–79. https://doi.org/10.1080/09629350220131917
- Marcel, A. K., Ekali, L. G., Eugene, S., Arnold, O. E., Sandrine, E. D., vor der Weid, D., Gbaguidi, E., Ngogang, J., & Mbanya, J. C. (2011). The effect of Spirulina platensis versus soybean on insulin resistance in HIV-infected patients: A randomized pilot study. Nutrients, 3(7), 712–724. https://doi.org/10.3390/nu3070712
- Cingi, C., Conk-Dalay, M., Cakli, H., & Bal, C. (2008). The effects of spirulina on allergic rhinitis. European Archives of Oto-Rhino-Laryngology, 265(10), 1219–1223. https://doi.org/10.1007/s00405-008-0642-8
- Park, H. J., Lee, Y. J., Ryu, H. K., Kim, M. H., Chung, H. W., & Kim, W. Y. (2008). A randomized double-blind, placebo-controlled study to establish the effects of spirulina in elderly Koreans. Annals of Nutrition and Metabolism, 52(4), 322–328. https://doi.org/10.1159/000151486
- Parikh, P., Mani, U., & Iyer, U. (2001). Role of Spirulina in the control of glycemia and lipidemia in type 2 diabetes mellitus. Journal of Medicinal Food, 4(4), 193–199. https://doi.org/10.1089/10966200152744463
- Torres-Duran, P. V., Ferreira-Hermosillo, A., & Juarez-Oropeza, M. A. (2007). Antihyperlipemic and antihypertensive effects of Spirulina maxima in an open sample of mexican population: A preliminary report. Lipids in Health and Disease, 6. https://doi.org/10.1186/1476-511X-6-33
- Lee, E. H., Park, J.-E., Choi, Y.-J., Huh, K.-B., & Kim, W.-Y. (2008). A randomized study to establish the effects of spirulina in type 2 diabetes mellitus patients. Nutrition Research and Practice, 2(4), 295. https://doi.org/10.4162/nrp.2008.2.4.295
- Kalafati, M., Jamurtas, A. Z., Nikolaidis, M. G., Paschalis, V., Theodorou, A. A., Sakellariou, G. K., Koutedakis, Y., & Kouretas, D. (2010). Ergogenic and antioxidant effects of spirulina supplementation in humans. Medicine and Science in Sports and Exercise, 42(1), 142–151. https://doi.org/10.1249/MSS.0b013e3181ac7a45
- Lu, H. K., Hsieh, C. C., Hsu, J. J., Yang, Y. K., & Chou, H. N. (2006). Preventive effects of Spirulina platensis on skeletal muscle damage under exercise-induced oxidative stress. European Journal of Applied Physiology, 98(2), 220–226. https://doi.org/10.1007/s00421-006-0263-0
- Dinicola, S., Pasqualato, A., Cucina, A., Coluccia, P., Ferranti, F., Canipari, R., Catizone, A., Proietti, S., D’Anselmi, F., Ricci, G., Palombo, A., & Bizzarri, M. (2014). Grape seed extract suppresses MDA-MB231 breast cancer cell migration and invasion. European Journal of Nutrition, 53(2), 421–431. https://doi.org/10.1007/s00394-013-0542-6
- Sano, A., Tokutake, S., & Seo, A. (2013). Proanthocyanidin-rich grape seed extract reduces leg swelling in healthy women during prolonged sitting. Journal of the Science of Food and Agriculture, 93(3), 457–462. https://doi.org/10.1002/jsfa.5773
- Sapwarobol, S., Adisakwattana, S., Changpeng, S., Ratanawachirin, W., Tanruttanawong, K., & Boonyarit, W. (2012). Postprandial blood glucose response to grape seed extract in healthy participants: A pilot study. Pharmacognosy Magazine, 8(31), 192–196. https://doi.org/10.4103/0973-1296.99283
- Olas, B., Wachowicz, B., Stochmal, A., & Oleszek, W. (2012). The polyphenol-rich extract from grape seeds inhibits platelet signaling pathways triggered by both proteolytic and non-proteolytic agonists. Platelets, 23(4), 282–289. https://doi.org/10.3109/09537104.2011.618562
- Yang, H., Lee, B. K., Kook, K. H., Jung, Y. S., & Ahn, J. (2012). Protective effect of grape seed extract against oxidative stress-induced cell death in a staurosporine-differentiated retinal ganglion cell line. Current Eye Research, 37(4), 339–344. https://doi.org/10.3109/02713683.2011.645106
- Raina, K., Tyagi, A., Kumar, D., Agarwal, R., & Agarwal, C. (2013). Role of oxidative stress in cytotoxicity of grape seed extract in human bladder cancer cells. Food and Chemical Toxicology, 61, 187–195. https://doi.org/10.1016/j.fct.2013.06.039
- Glurich, I., Grossi, S., Albini, B., Ho, A., Shah, R., Zeid, M., Baumann, H., Genco, R. J., & De Nardin, E. (2002). Systemic inflammation in cardiovascular and periodontal disease: Comparative study. Clinical and Diagnostic Laboratory Immunology, 9(2), 425–432. https://doi.org/10.1128/CDLI.9.2.425-432.2002
- Kimmatkar, N., Thawani, V., Hingorani, L., & Khiyani, R. (2003). Efficacy and tolerability of Boswellia serrata extract in treatment of osteoarthritis of knee - A randomized double blind placebo controlled trial. Phytomedicine, 10(1), 3–7. https://doi.org/10.1078/094471103321648593
- Sailer, E. R., Subramanian, L. R., Rall, B., Hoernlein, R. F., Ammon, H. P. T., & Safayhi, H. (1996). Acetyl-11-keto-β-boswellic acid (AKBA): Structure requirements for binding and 5-lipoxygenase inhibitory activity. British Journal of Pharmacology, 117(4), 615–618. https://doi.org/10.1111/j.1476-5381.1996.tb15235.x
- Siddiqui, M. Z. (2011). Boswellia serrata, a potential antiinflammatory agent: An overview. In Indian Journal of Pharmaceutical Sciences (Vol. 73, Issue 3, pp. 255–261). Indian J Pharm Sci. https://doi.org/10.4103/0250-474X.93507
- Bayrak, Ş., & Mitchison, N. A. (1998). Bystander suppression of murine collagen-induced arthritis by long-term nasal administration of a self type II collagen peptide. Clinical and Experimental Immunology, 113(1), 92–95. https://doi.org/10.1046/j.1365-2249.1998.00638.x
- Lugo, J. P., Saiyed, Z. M., Lau, F. C., Molina, J. P. L., Pakdaman, M. N., Shamie, A. N., & Udani, J. K. (2013). Undenatured type II collagen (UC-II®) for joint support: A randomized, double-blind, placebo-controlled study in healthy volunteers. Journal of the International Society of Sports Nutrition, 10, 48. https://doi.org/10.1186/1550-2783-10-48
- Nagler-Anderson, C., Bober, L. A., Robinson, M. E., Siskind, G. W., & Thorbecke, G. J. (1986). Suppression of type II collagen-induced arthritis by intragastric administration of soluble type II collagen. Proceedings of the National Academy of Sciences of the United States of America, 83(19), 7443–7446. https://doi.org/10.1073/pnas.83.19.7443
- Jackson, J. K., Higo, T., Hunter, W. L., & Burt, H. M. (2006). The antioxidants curcumin and quercetin inhibit inflammatory processes associated with arthritis. Inflammation Research, 55(4), 168–175. https://doi.org/10.1007/s00011-006-0067-z
- Shoba, G., Joy, D., Joseph, T., Majeed, M., Rajendran, R., & Srinivas, P. S. S. R. (1998). Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Medica, 64(4), 353–356. https://doi.org/10.1055/s-2006-957450
- Sharma, R. A., Euden, S. A., Platton, S. L., Cooke, D. N., Shafayat, A., Hewitt, H. R., Marczylo, T. H., Morgan, B., Hemingway, D., Plummer, S. M., Pirmohamed, M., Gescher, A. J., & Steward, W. P. (2004). Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clinical Cancer Research, 10(20), 6847–6854. https://doi.org/10.1158/1078-0432.CCR-04-0744
- Anand, P., Kunnumakkara, A. B., Newman, R. A., & Aggarwal, B. B. (2007). Bioavailability of curcumin: Problems and promises. In Molecular Pharmaceutics (Vol. 4, Issue 6, pp. 807–818). Mol Pharm. https://doi.org/10.1021/mp700113r
- Aggarwal, S., Ichikawa, H., Takada, Y., Sandur, S. K., Shishodia, S., & Aggarwal, B. B. (2006). Curcumin (diferuloylmethane) down-regulates expression of cell proliferation and antiapoptotic and metastatic gene products through suppression of IκBα kinase and Akt activation. Molecular Pharmacology, 69(1), 195–206. https://doi.org/10.1124/mol.105.017400
- Aggarwal, B. B., Sundaram, C., Malani, N., & Ichikawa, H. (2007). Curcumin: The Indian solid gold. In Advances in Experimental Medicine and Biology (Vol. 595, pp. 1–75). Springer New York LLC. https://doi.org/10.1007/978-0-387-46401-5_1
- Brien, S., Prescott, P., Bashir, N., Lewith, H., & Lewith, G. (2008). Systematic review of the nutritional supplements dimethyl sulfoxide (DMSO) and methylsulfonylmethane (MSM) in the treatment of osteoarthritis. In Osteoarthritis and Cartilage (Vol. 16, Issue 11, pp. 1277–1288). Osteoarthritis Cartilage. https://doi.org/10.1016/j.joca.2008.03.002
- Reichenbach, S., Sterchi, R., Scherer, M., Trelle, S., Bürgi, E., Bürgi, U., Dieppe, P. A., & Jüni, P. (2007). Meta-analysis: Chondroitin for osteoarthritis of the knee or hip. In Annals of Internal Medicine (Vol. 146, Issue 8, pp. 580–590). American College of Physicians. https://doi.org/10.7326/0003-4819-146-8-200704170-00009
- McAlindon, T. E., La Valley, M. P., Gulin, J. P., & Felson, D. T. (2000). Glucosamine and chondroitin for treatment of osteoarthritis: A systematic quality assessment and meta-analysis. In Journal of the American Medical Association (Vol. 283, Issue 11, pp. 1469–1475). American Medical Association. https://doi.org/10.1001/jama.283.11.1469
- Wandel, S., Jüni, P., Tendal, B., Nüesch, E., Villiger, P. M., Welton, N. J., Reichenbach, S., & Trelle, S. (2010). Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: Network meta-analysis. BMJ (Online), 341(7775), 711. https://doi.org/10.1136/bmj.c4675
- Momomura, R., Naito, K., Igarashi, M., Watari, T., Terakado, A., Oike, S., Sakamoto, K., Nagaoka, I., & Kaneko, K. (2013). Evaluation of the effect of glucosamine administration on biomarkers of cartilage and bone metabolism in bicycle racers. Molecular Medicine Reports, 7(3), 742–746. https://doi.org/10.3892/mmr.2013.1289
- Herrero-Beaumont, G., Román Ivorra, J. A., Trabado, M. D. C., Blanco, F. J., Benito, P., Martìn-Mola, E., Paulino, J., Marenco, J. L., Porto, A., Laffon, A., Araújo, D., Figueroa, M., & Branco, J. (2007). Glucosamine sulfate in the treatment of knee osteoarthritis symptoms: A randomized, double-blind, placebo-controlled study using acetaminophen as a side comparator. Arthritis and Rheumatism, 56(2), 555–567. https://doi.org/10.1002/art.22371
- Widrig, R., Suter, A., Saller, R., & Melzer, J. (2007). Choosing between NSAID and arnica for topical treatment of hand osteoarthritis in a randomised, double-blind study. Rheumatology International, 27(6), 585–591. https://doi.org/10.1007/s00296-007-0304-y
- Knuesel, O., Weber, M., & Suter, A. (2002). Arnica montana gel in osteoarthritis of the knee: An open, multicenter clinical trial. Advances in Therapy, 19(5), 209–218. https://doi.org/10.1007/BF02850361
- Kong, L. J., Zhan, H. S., Cheng, Y. W., Yuan, W. A., Chen, B., & Fang, M. (2013). Massage therapy for neck and shoulder pain: A systematic review and meta-analysis. In Evidence-based Complementary and Alternative Medicine (Vol. 2013). Evid Based Complement Alternat Med. https://doi.org/10.1155/2013/613279
- Liu, L., Huang, Q. M., Liu, Q. G., Ye, G., Bo, C. Z., Chen, M. J., & Li, P. (2015). Effectiveness of dry needling for myofascial trigger points associated with neck and shoulder pain: A systematic review and meta-analysis. In Archives of Physical Medicine and Rehabilitation (Vol. 96, Issue 5, pp. 944–955). W.B. Saunders. https://doi.org/10.1016/j.apmr.2014.12.015
- Noten, S., Meeus, M., Stassijns, G., Van Glabbeek, F., Verborgt, O., & Struyf, F. (2016). Efficacy of Different Types of Mobilization Techniques in Patients with Primary Adhesive Capsulitis of the Shoulder: A Systematic Review. In Archives of Physical Medicine and Rehabilitation (Vol. 97, Issue 5, pp. 815–825). W.B. Saunders. https://doi.org/10.1016/j.apmr.2015.07.025
- Zheng, X. Q., Li, K., Wei, Y. D., Tie, H. T., Yi, X. Y., & Huang, W. (2014). Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: A systematic review and meta-analysis. In Archives of Physical Medicine and Rehabilitation (Vol. 95, Issue 10, pp. 1824–1831). W.B. Saunders. https://doi.org/10.1016/j.apmr.2014.04.024
- Requa, R. K., Deavilla, L. N., & Garrick, J. G. (1993). Injuries in recreational adult fitness activities. The American Journal of Sports Medicine, 21(3), 461–467. https://doi.org/10.1177/036354659302100323
- Brooks, J. H. M., Fuller, C. W., Kemp, S. P. T., & Reddin, D. B. (2006). Incidence, risk, and prevention of hamstring muscle injuries in professional rugby union. American Journal of Sports Medicine, 34(8), 1297–1306. https://doi.org/10.1177/0363546505286022
- Kerr, Z. Y., Collins, C. L., & Dawn Comstock, R. (2010). Epidemiology of Weight Training-Related Injuries Presenting to United States Emergency Departments, 1990 to 2007. In American Journal of Sports Medicine (Vol. 38, Issue 4, pp. 765–771). SAGE Publications Inc. https://doi.org/10.1177/0363546509351560
- Colado, J. C., Pablos, C., Chulvi-Medrano, I., Garcia-Masso, X., Flandez, J., & Behm, D. G. (2011). The progression of paraspinal muscle recruitment intensity in localized and global strength training exercises is not based on instability alone. Archives of Physical Medicine and Rehabilitation, 92(11), 1875–1883. https://doi.org/10.1016/j.apmr.2011.05.015
- Cholewicki, J., & McGill, S. M. (1992). Lumbar posterior ligament involvement during extremely heavy lifts estimated from fluoroscopic measurements. Journal of Biomechanics, 25(1), 17–28. https://doi.org/10.1016/0021-9290(92)90242-S
- Uthman, O. A., Van Der Windt, D. A., Jordan, J. L., Dziedzic, K. S., Healey, E. L., Peat, G. M., & Foster, N. E. (2013). Exercise for lower limb osteoarthritis: Systematic review incorporating trial sequential analysis and network meta-analysis. BMJ (Online), 347(7928). https://doi.org/10.1136/bmj.f5555
- Toutoungi, D. E., Lu, T. W., Leardini, A., Catani, F., & O’Connor, J. J. (2000). Cruciate ligament forces in the human knee during rehabilitation exercises. Clinical Biomechanics, 15(3), 176–187. https://doi.org/10.1016/S0268-0033(99)00063-7
- Nagura, T., Dyrby, C. O., Alexander, E. J., & Andriacchi, T. P. (2002). Mechanical loads at the knee joint during deep flexion. Journal of Orthopaedic Research, 20(4), 881–886. https://doi.org/10.1016/S0736-0266(01)00178-4
- B. FITZGERALD, F. (G)., & G. R. MOLATCHIE, F. (G). . (n.d.). DEGENERATIVE JOINT DISEASE IN WEIGHT-LIFTERS FACTOR FICTION? Retrieved November 12, 2020, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1858976/pdf/brjsmed00258-0029.pdf
- Crowley, D. C., Lau, F. C., Sharma, P., Evans, M., Guthrie, N., Bagchi, M., Bagchi, D., Dey, D. K., & Raychaudhuri, S. P. (2009). Safety and efficacy of undenatured type II collagen in the treatment of osteoarthritis of the knee: A clinical trial. International Journal of Medical Sciences, 6(6), 312–321. https://doi.org/10.7150/ijms.6.312
- Deparle, L. A., Gupta, R. C., Canerdy, T. D., Goad, J. T., D’Altilio, M., Bagchi, M., & Bagchi, D. (2005). Efficacy and safety of glycosylated undenatured type-II collagen (UC-II) in therapy of arthritic dogs. Journal of Veterinary Pharmacology and Therapeutics, 28(4), 385–390. https://doi.org/10.1111/j.1365-2885.2005.00668.x
- Chakravarty, E. F., Hubert, H. B., Lingala, V. B., Zatarain, E., & Fries, J. F. (2008). Long Distance Running and Knee Osteoarthritis. A Prospective Study. American Journal of Preventive Medicine, 35(2), 133–138. https://doi.org/10.1016/j.amepre.2008.03.032
- Eckstein, F., Hudelmaier, M., & Putz, R. (2006). The effects of exercise on human articular cartilage. In Journal of Anatomy (Vol. 208, Issue 4, pp. 491–512). J Anat. https://doi.org/10.1111/j.1469-7580.2006.00546.x
- Adams, M. A., & Dolan, P. (1995). Recent advances in lumbar spinal mechanics and their clinical significance. In Clinical Biomechanics (Vol. 10, Issue 1, pp. 3–19). Clin Biomech (Bristol, Avon). https://doi.org/10.1016/0268-0033(95)90432-9