Retatrutide, or “reta,” is an experimental weight-loss drug similar to other weight-loss drugs like semaglutide and tirzepatide.
It’s a once-weekly injection designed to help people lose weight, improve blood sugar control, and reduce problems linked to excess body fat, such as fatty liver disease, high blood pressure, and abnormal cholesterol.
Retatrutide is currently in Phase 3 clinical trials. That means it’s passed earlier tests of basic safety and effectiveness, and researchers are now testing it in larger groups to confirm how well it works and whether it’s safe enough for doctors to prescribe.
Until the FDA reviews the full evidence and approves retatrutide, it won’t be legally available as a prescription drug and won’t have an official dose, price, or label.
Table of Contents
+
Key Takeaways
- Retatrutide mimics three hormones—GLP-1, GIP, and glucagon—which helps people eat less and lose weight.
- Early studies suggest retatrutide may cause more weight loss than semaglutide and tirzepatide, with some people losing 30% of their body weight or more.
- Retatrutide is not FDA approved, isn’t legally available by prescription, and may cause stronger side effects at higher doses.
How Does Retatrutide Work?
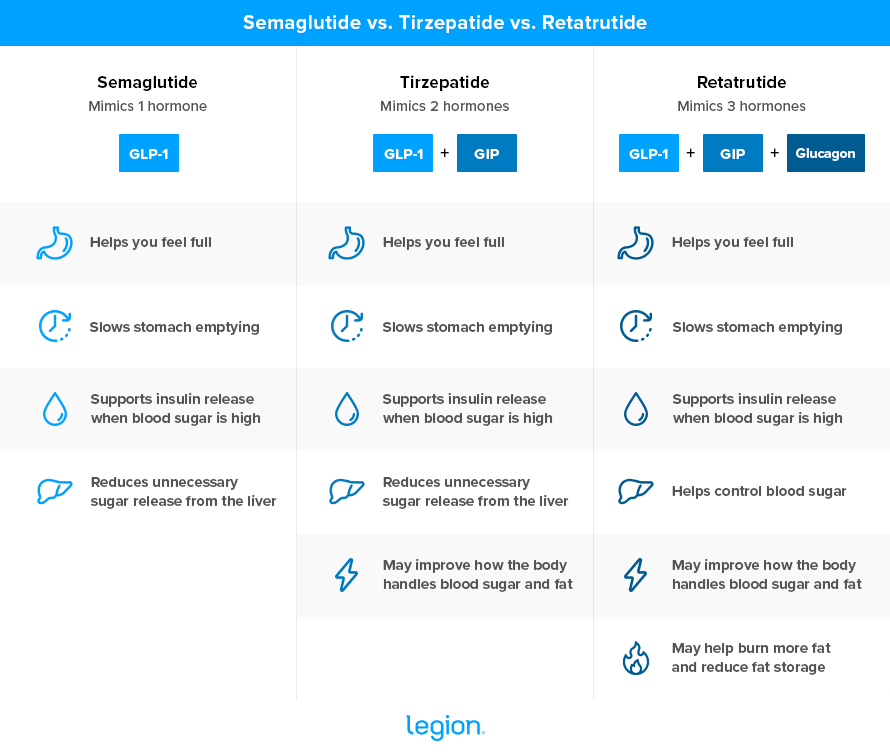
Retatrutide is a peptide that mimics three hormones in the body: GLP-1, GIP, and glucagon
Your gut releases GLP-1 after you eat, and it helps you feel full, slows how quickly food leaves your stomach, tells your pancreas to release insulin when blood sugar is high, and reduces how much sugar your liver releases into your blood.
Like GLP-1, GIP helps your pancreas release insulin after meals, which helps move sugar out of your blood and into your cells.
Glucagon helps control how your body handles stored energy. It tells your liver to send stored sugar into your blood, and it may also help your body make less new fat and burn more stored fat. Researchers think this extra action may help retatrutide cause more fat loss than drugs that mimic only GLP-1, or GLP-1 plus GIP.
The two most popular weight loss drugs—semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound)—target GLP-1 alone or GLP-1 plus GIP, but not glucagon. Retatrutide targets all three, theoretically making it the most potent weight loss drug in its class.
Here’s how the three drugs compare at a glance:
How Much Weight Can You Lose With Retatrutide?
Early studies show that people taking retatrutide usually lose around 15–30% of their body weight, with some losing substantially more. Here’s what the results look like compared to semaglutide and tizepatide:
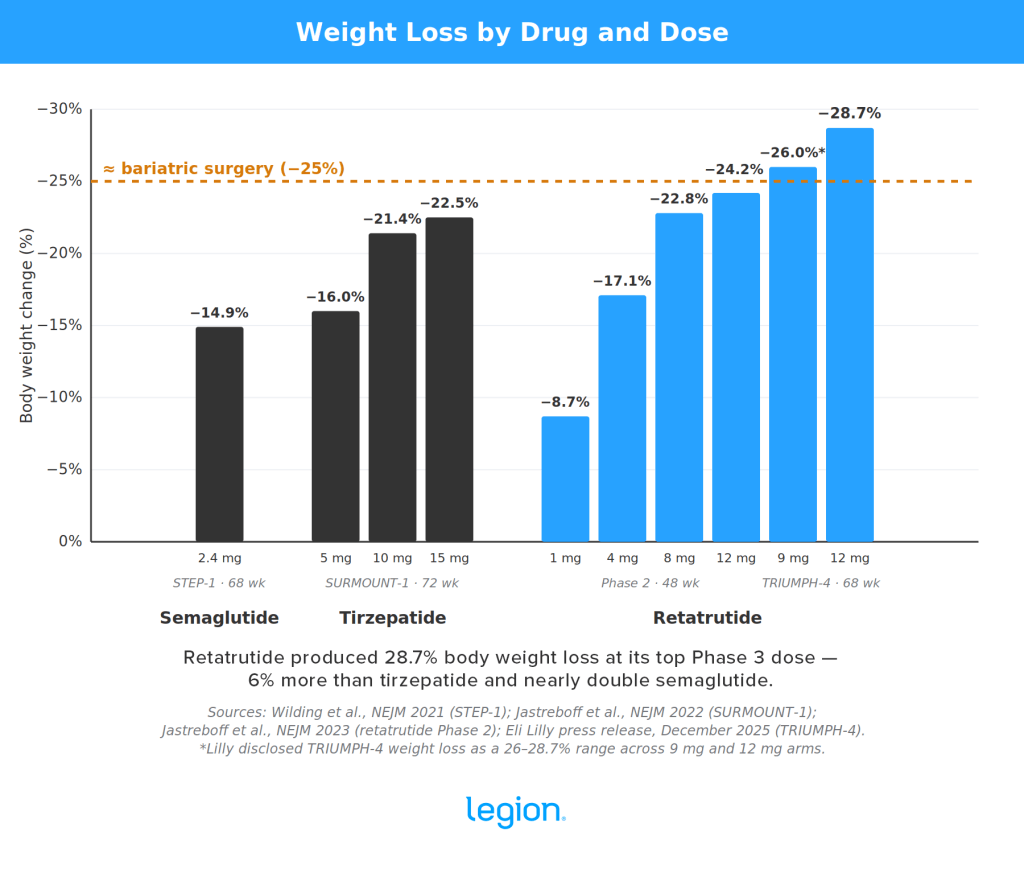
In a 2023 study by Yale University School of Medicine, researchers gave 338 adults a weekly injection of retatrutide or placebo for 48 weeks. People taking the highest dose of retatrutide lost just over 24% of their body weight, and almost a quarter of the participants lost 30% of their body weight. People taking a placebo lost around 2%.1
Then, in 2025, the drug’s manufacturer—Eli Lilly—announced preliminary results from a Phase 3 trial of 445 adults.2 People taking the highest dose of retatrutide lost just under 29% of their body weight after 68 weeks, compared with 2% in the placebo group.
For a 250-pound person, that would mean losing around 72 pounds in about 16 months.
The same trial found that 39% of people taking the highest dose lost 30% or more of their body weight, and 24% lost 35% or more. These results haven’t been published in a peer-reviewed journal yet, but they’re the most up-to-date retatrutide weight-loss data available.
For comparison, the average bariatric surgery patient loses about 25-to-35% of their body weight. In other words, retatrutide causes comparable weight loss to what occurs after surgically altering the size and function of your stomach.
A 2025 meta-analysis by South Valley University found a similar pattern when it pooled the Yale trial with 2 others. Across the three studies, retatrutide reduced body weight by an average of 14 percentage points more than placebo after 36 weeks.3
The meta-analysis also found that higher doses generally caused more weight loss. Compared with placebo, retatrutide led to 10 percentage points more weight loss with 4 mg, 16 points with 8 mg, and 17 points with 12 mg.
These are incredible early results, but so far, only a handful of studies have tested retatrutide and it still hasn’t received FDA approval for weight loss.
What Else Does Retatrutide Do?
In addition to weight loss, retatrutide reduces …
- Blood sugar. In a study from the National Cancer Institute, retatrutide reduced HbA1c (a marker of long-term blood sugar levels) by 2% after 36 weeks.4 For comparison, metformin—a powerful diabetes drug—typically drops HbA1c by about 1 to 1.5%, and high-dose semaglutide (Ozempic) reduces it by about 1.5 to 1.8%.
- Liver fat. In a study from Virginia Commonwealth University, 12 mg of retatrutide reduced liver fat by 82% after a year, with about 80% of participants reaching healthy levels of liver fat.5
- Cholesterol and triglycerides. Across all trials on retatrutide thus far, LDL dropped about 10–15% and triglycerides dropped 20–30% at higher doses.
- Blood pressure. Several studies have found that systolic blood pressure dropped roughly 7 to 10 mmHg at higher doses of retatrutide—similar to or slightly better than what typically occurs on tirzepatide.67
All of that said, these benefits are primarily due to the weight loss itself rather than retatrutide causing them directly. That is, the more weight people lose, the more of a reduction in blood sugar, liver fat, blood lipids, and blood pressure they experience. Still, the benefits are on par with other forms of rapid weight loss and worth highlighting.
What Are Retatrutide’s Side Effects?
Retatrutide’s most common side effects are stomach-related problems. In the Yale study, the most commonly reported issues were nausea, diarrhea, vomiting, and constipation.
These side effects were usually worse at higher doses. For example, nausea affected 45% of people taking the highest dose of retatrutide, compared with 11% of people taking placebo. Vomiting affected 19% of people taking the highest dose, compared with 1% taking placebo.
Eli Lilly reported a similar pattern in its Phase 3 study results. At the highest dose, nausea affected 43% of people, diarrhea affected 33%, constipation affected 25%, and vomiting affected 21%. Twenty-one percent of those taking the high dose also reported abnormal skin sensations, such as tingling, burning, or sensitivity.
For some people, these side effects were too rough to continue treatment. In the highest-dose group, 16% of people stopped taking retatrutide because of side effects, while no one in the placebo group stopped for this reason.
For context, side effects caused about 5% of people to stop semaglutide and about 7% to stop tirzepatide in similar weight-loss trials.89
The Yale study found that retatrutide also raised heart rate. The increase was larger at higher doses, peaked after 24 weeks of treatment, then went down later in the trial.
In other words, retatrutide’s side effects look similar to other weight-loss drugs. At higher doses, it may be harder to tolerate, but we won’t fully understand its long-term safety risks until we have more data.
What Is the Dosage of Retatrutide?
Scientists are still trying to establish the most effective dose of retatrutide. Any dose you see online is based on preliminary trials, not an FDA-approved dose.
In the Yale study, researchers tested once-weekly injections of 1 mg, 4 mg, 8 mg, and 12 mg for 48 weeks. People assigned to the higher-dose groups usually started lower, then increased their dose every 4 weeks to make the drug easier to tolerate.
In Eli Lilly’s Phase 3 study, researchers tested 9 mg and 12 mg. Participants started at 2 mg once per week and increased the dose every 4 weeks until they reached their target dose.
In both studies, 12 mg produced the most weight loss, but that doesn’t mean it’s the “right” dose. Higher doses also tended to be harder to tolerate.
The 2025 South Valley University meta-analysis found the same tradeoff: higher doses generally caused more weight loss, but they also increased the risk of side effects. Compared with placebo, side effects weren’t significantly higher with 4 mg, but they were higher with 8 mg and 12 mg.
Is Retatrutide Better Than Tirzepatide and Semaglutide?
Retatrutide may cause more weight loss than tirzepatide and semaglutide, but it’s too early to say it’s better overall. Both tirzepatide and semaglutide are FDA-approved, have support from more long-term data, and are already used in real-life settings. Retatrutide, on the other hand, is promising but still very much experimental.
What’s more, the early data indicates that side effects are more common and severe with retatrutide than either tirzepatide or semaglutide.
Here’s how they compare at a glance:
Is Retatrutide Better Than Tirzepatide?
| Category | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Drug type | Single agonist | Dual agonist | Triple agonist |
| Hormones mimicked | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + glucagon |
| FDA status | Approved for weight loss and type 2 diabetes | Approved for weight loss and type 2 diabetes | Not approved; still in trials |
| Dosing | FDA-approved dosing instructions exist | FDA-approved dosing instructions exist | No official dose yet |
| Evidence | Strong Phase 3 evidence and widespread real-world use | Strong Phase 3 evidence plus growing real-world data | Strong Phase 2 evidence plus early Phase 3 topline data |
| Average weight loss | About 15% in major obesity trials | Around 20% or more in major obesity trials | Up to just under 29% in Phase 3 topline results |
| Speed of weight loss | Large losses, usually measured over about 68 weeks | Large losses, usually measured over about 72 weeks | Very large losses by 48–68 weeks in early trials |
| Side effects | Mainly nausea, vomiting, diarrhea, and constipation | Mainly nausea, vomiting, diarrhea, and constipation | Mainly nausea, vomiting, diarrhea, constipation, and abnormal skin sensations |
| Tolerability | Side effects caused about 5% of people to stop in a major weight-loss trial | Side effects caused about 7% of people to stop in a major weight-loss trial | Side effects caused 16–18% of people on the highest dose to stop in early trials |
| Practical takeaway | Proven, approved, and widely used | More powerful than semaglutide and already approved | Potentially most powerful, but still experimental |

Where Can You Get Retatrutide?
The only legitimate way to get retatrutide at the moment is through a clinical trial. That means you’d have to qualify for a study, be accepted by the research team, and take the drug under medical supervision.
You can’t get retatrutide from a regular doctor or pharmacy because scientists are still studying it, so it’s not available on prescription like Ozempic, Wegovy, or Mounjaro.
This is where things get messy.
Plenty of websites already sell “retatrutide peptides,” but these aren’t FDA-approved medications. You have no way to know whether they contain an effective dose of real retatrutide, or if they’re safe—which is why the FDA strongly recommends against purchasing them.10
That doesn’t mean every vial sold online is definitely fake or tainted. It means you’re gambling with a drug that hasn’t been approved, from sellers who don’t have to meet the same standards as real drug manufacturers.
The most sensible option right now is to skip the online retatrutide peptides sellers and stick with other effective, safe, and legally available options. If retatrutide works as well as early trials suggest, it shouldn’t be too long to wait for FDA review and a real prescription version.
FAQ #1: Does retatrutide actually burn fat?
Retatrutide could help your body burn more fat because it mimics glucagon, a hormone involved in how your body uses stored energy.11 When glucagon levels rise, the liver tends to make less new fat and hold on to less fat overall. It may also increase calorie burning, though the human evidence for this is still mixed.
In other words, retatrutide isn’t a “fat burner” in the supplement-marketing sense. It helps you lose fat by tanking your appetite, which allows you to eat less. It may slightly increase the percentage of weight you lose that’s fat, but the evidence isn’t strong enough to show this conclusively.
FAQ #2: Is retatrutide FDA approved?
No, retatrutide is not FDA approved. It’s still being tested in Phase 3 clinical trials, which are the large studies researchers run before asking regulators to approve a new drug. Until the FDA reviews the full evidence and approves it, retatrutide won’t be legally available as a prescription drug.
FAQ #3: Who should not take retatrutide?
No one should take retatrutide right now unless they’re in a supervised clinical trial.
Retatrutide is still experimental, so doctors don’t yet have official FDA guidance on who can use it safely and who should avoid it. If anyone gives a more specific answer than that, it’s an educated guess based on early trial data and what we know about similar drugs.
Want More Content Like This?
Check out these articles:
- Do GLP-1s Actually Cause Muscle Loss?
- What Happens When You Stop Taking Ozempic?
- An Evidence-Based Guide to 7 Popular Weight Loss Pills and Injections
Scientific References +
- ↩ Jastreboff AM, Kaplan LM, Frías JP, et al. Triple-hormone-receptor agonist retatrutide for obesity – a phase 2 trial. N Engl J Med. 2023;389(6):514-526.
- ↩ Eli Lilly and Company. Lilly’s triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial. December 2025.
- ↩ Abouelmagd AA, Abdelrehim AM, Bashir MN, et al. Efficacy and safety of retatrutide, a novel GLP-1, GIP, and glucagon receptor agonist for obesity treatment: a systematic review and meta-analysis of randomized controlled trials. Proc (Bayl Univ Med Cent). 2025;38(3):291-303.
- ↩ Rosenstock J, Frias J, Jastreboff AM, et al. Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet. 2023;402(10401):529-544.
- ↩ Sanyal AJ, Kaplan LM, Frias JP, et al. Triple hormone receptor agonist retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomized phase 2a trial. Nat Med. 2024;30(7):2037-2048.
- ↩ Jastreboff AM, Kaplan LM, Frías JP, et al. Triple-hormone-receptor agonist retatrutide for obesity – a phase 2 trial. N Engl J Med. 2023;389(6):514-526.
- ↩ Eli Lilly and Company. Lilly’s triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial. December 2025.
- ↩ Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
- ↩ Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216.
- ↩ U.S. Food and Drug Administration. FDA’s concerns with unapproved GLP-1 drugs used for weight loss. Accessed May 2026.
- ↩ Newsome PN, Ambery P. Incretins (GLP-1 receptor agonists and dual/triple agonists) and the liver. J Hepatol. 2023;79(6):1557-1565.